To overcome this problem and to increase the resolving power of this technique a single sample can be digested in parallel by several enzymes (often three) resulting in three T-RFLP profiles per sample each resolving some variants while missing others. Often, several different bacteria in a population might give a single peak on the electropherogram due to the presence of a restriction site for the particular restriction enzyme used in the experiment at the same position. Both assumptions listed above, however, are not always met. 
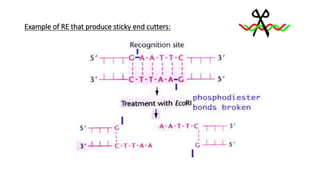
In a T–RFLP profile each peak assumingly corresponds to one genetic variant in the original sample while its height or area corresponds to its relative abundance in the specific community. Thus, what appears on an electrophoresis gel as a band appears as a peak on the electropherogram whose integral is its total fluorescence. In an electropherogram the X-axis marks the sizes of the fragments while the Y-axis marks the fluorescence intensity of each fragment. The result of a T-RFLP profiling is a graph called electropherogram which is an intensity plot representation of an electrophoresis experiment (gel or capillary). In addition to these steps the TRFLP protocol often includes a cleanup of the PCR products prior to the restriction and in case a capillary electrophoresis is used a desalting stage is also performed prior to running the sample. Thus, T-RFLP is different from ARDRA and RFLP in which all restriction fragments are visualized. the labeled end or ends of the amplicon) are read while all other fragments are ignored. Because the excised mixture of amplicons is analyzed in a sequencer, only the terminal fragments (i.e. Following the restriction reaction, the mixture of fragments is separated using either capillary or polyacrylamide electrophoresis in a DNA sequencer and the sizes of the different terminal fragments are determined by the fluorescence detector. The mixture of amplicons is then subjected to a restriction reaction, normally using a four-cutter restriction enzyme. While several common fluorescent dyes can be used for the purpose of tagging such as 6-carboxyfluorescein (6-FAM), ROX, carboxytetramethylrhodamine (TAMRA, a rhodamine-based dye), and hexachlorofluorescein (HEX), the most widely used dye is 6-FAM. In case both primers are labeled, different fluorescent dyes are required. In the case of TRFLP, the amplification is performed with one or both the primers having their 5’ end labeled with a fluorescent molecule. Like most other community analysis methods, TRFLP is also based on PCR amplification of a target gene. Since then the method has been applied for the use of other marker genes such as the functional marker gene pmoA to analyze methanotrophic communities. The method was first described by Avaniss-Aghajani et al in 1994 and later by Liu in 1997 which employed the amplification of the 16S rDNA target gene from the DNA of several isolated bacteria as well as environmental samples. These relatively high throughput methods were developed in order to reduce the cost and effort in analyzing microbial communities using a clone library. Other similar methods include DGGE, TGGE, ARISA, ARDRA, PLFA, etc. 
TRFLP is one of several molecular methods aimed to generate a fingerprint of an unknown microbial community. The result is a graph image where the x-axis represents the sizes of the fragment and the y-axis represents their fluorescence intensity. The method is based on digesting a mixture of PCR amplified variants of a single gene using one or more restriction enzymes and detecting the size of each of the individual resulting terminal fragments using a DNA sequencer. Terminal restriction fragment length polymorphism ( TRFLP or sometimes T-RFLP) is a molecular biology technique for profiling of microbial communities based on the position of a restriction site closest to a labelled end of an amplified gene. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
